Boston Food Allergy Center
To help patients with food related disorders, Dr. Leung established Boston Food Allergy Center in 2015 with the goal to treat these conditions using a multidisciplinary approach: GI + allergy + nutrition. Having fellowship-trained in both allergy and gastroenterology subspecialties, Dr. Leung has unique knowledge and experience to evaluate patients from the perspectives of both an allergist and a gastroenterologist in the same setting.
Since 2015, we have grown our team from only 3 members to now over 20. We have expanded our clinic space twice, from a small office to now 2 offices occupying over 3000 sq ft in downtown Boston. The expansion of both physical space and our team allows us to take care of patients with non-food related common allergy and GI conditions. In 2021, we decided to change our name to Boston Specialists to reflect our expanded scope of practice.
Dr. Leung is board certified for both Gastroenterology and Allergy/Immunology. We treat hundreds of pediatric and adult patients with food allergy, eosinophilic esophagitis (EoE), eosinophilic gastrointestinal disorders, mast cell activation related GI conditions, food protein induced enterocolitis syndrome (FPIES) and other food related diseases every month.
We are among the first to offer oral immunotherapy (OIT) as a therapeutic option to treat patients with severe food allergy. Hundreds of patients have successfully undergone single/multiple-food OIT since 2017. Peanuts, tree nuts, chickpea, sesame, egg and milk are some of the examples of food we use for OIT. Our team is also experienced in using elimination diet to treat a variety of food related disorders such as EoE and food intolerance.
Conditions we treat
Alpha-gal
Anaphylaxis
Angiodema/swelling
Burning mouth syndrome
Celiac disease
Eosinophilic Esophagitis
Eosinophilic Gastrointestinal Disorders
Food allergy and oral immunotherapy
Food dependent exercise induced anaphylaxis
Food independent, exercise induced anaphylaxis
Food-induced inflammatory disorders
Food Intolerance (Lactose, Fructose, Fructan, Sorbitol and others)
Food protein induced enterocolitis syndrome (FPIES)
Hives (Urticaria)
Mast Cell Activation Disorder
Oral allergy syndrome
Pruritus
Wheat hypersensitivity/
Non-Celiac
Diagnostic/Instructions
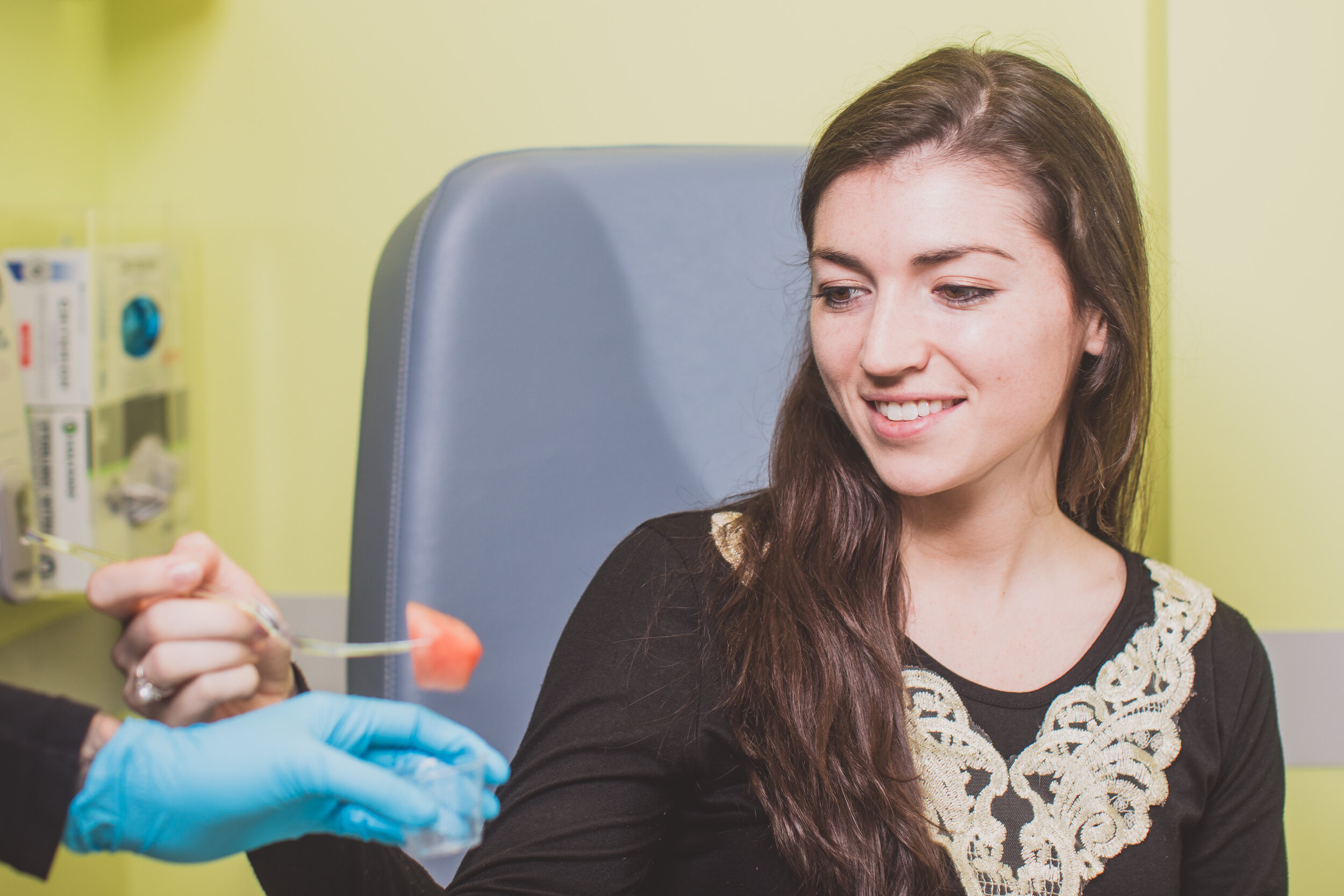
We offer some of the most comprehensive state-of-the-art diagnostic studies for food allergies at our center. This includes skin prick testing, supervised oral food challenge, component-resolved diagnostics, oral immunotherapy (OIT), and endoscopies etc. We have in-house laboratory service provided by Quest Diagnostics and we have a nutritionist on-site to address any nutritional concerns.